 Modern healthcare is undergoing a radical transformation. Medical care for high-output equipment is rapidly migrating from sterile clinical wards directly into the patient’s home. This shift creates a unique engineering hurdle: devices like portable surgical pumps and diagnostic monitors, once tucked away in noisy hospital wings, must now meet consumer-grade ergonomic and comfort standards to ensure patient compliance in a home environment.
Modern healthcare is undergoing a radical transformation. Medical care for high-output equipment is rapidly migrating from sterile clinical wards directly into the patient’s home. This shift creates a unique engineering hurdle: devices like portable surgical pumps and diagnostic monitors, once tucked away in noisy hospital wings, must now meet consumer-grade ergonomic and comfort standards to ensure patient compliance in a home environment.
While internal noise reduction is vital, the external interface often dictates the patient’s physical comfort and the device’s actual usability. To succeed in this market, Original Equipment Manufacturers (OEMs) must move beyond basic utility and prioritize a touchpoint-first philosophy using advanced molded solutions like POLYFORM®.
At Polymer, we believe that the physical touchpoint is a critical component of care. By utilizing advanced materials such as POLYFORM®, we help engineers bridge the gap between high-performance diagnostics and the tactile comfort required for daily home use.
In this blog article, we will explore:
- How POLYFORM® custom molding addresses tactile comfort and ergonomic challenges.
- The role of Indention Force Deflection (IFD) in finding the perfect level of firmness for patients.
- A case study on a heart monitor interface that improved both manufacturability and patient experience.
- How molded foam provides integrated protection for sensitive electronics and EMI/RFI shielding.
Why POLYFORM® is the Key to Home-Ready Design
While materials like POLYDAMP® Melamine Foam (PMF) are excellent for internal acoustical absorption and thermal management, POLYFORM® custom molded foam allows engineers to address the external patient interface with precision.
Tactile Customization
Using Indention Force Deflection (IFD) standards, engineers can utilize POLYFORM® Low Density Molded—High Resilient (PLDM-HR) polyurethane foam to find the exact level of firmness required for patient comfort.
Integrated Protection
The molding process allows for sensitive electronics to be encased deep inside the foam cushion rather than being exposed, protecting the brain of the device from external wear.
Design Efficiency
By switching to a custom molded POLYFORM® solution, manufacturers can simplify assembly and create products that are more cost-competitive than those using traditional multi-part suppliers.
A case study: Prioritizing external patient touchpoints
While internal engineering manages technical performance, the external interface dictates the patient’s physical comfort and the device’s actual usability. This is especially critical for next-generation medical devices where patients interact directly with the equipment for extended periods.
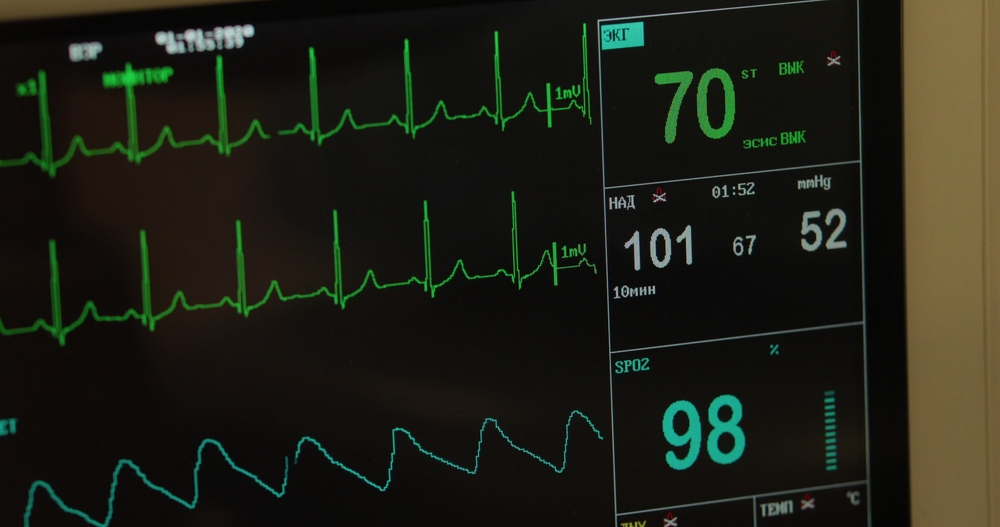
Perfecting the heart monitor interface
Polymer Technologies recently collaborated with a medical manufacturer to redesign a heart monitoring device that highlights the importance of this touchpoint-first philosophy. The system consisted of two components: a device placed inside the patient’s heart and a supportive foam cushion containing the electronics that read the data.
Because patients were required to lie directly on the cushion, the first-generation design faced significant challenges regarding comfort and cost-effectiveness. This particular project focused on several key improvements:
- Tactile customization via IFD testing – Using Indention Force Deflection (IFD) standards, engineers tested until they found the precise level of firmness required for patient comfort.
- The PLDM-HR solution – Our team selected POLYFORM® Low Density Molded—High Resilient (PLDM-HR) polyurethane foam.
- Integrated protective padding – The design encased the sensitive electronics deep inside the foam cushion rather than exposing them.
- EMI/RFI shielding for data integrity – To prevent home electronics or WiFi from interfering with the heart data, a conductive fabric shield was added to the back of the foam cushion.
Engineering for efficiency and comfort
By switching to a custom molded solution, the manufacturer didn’t just improve the patient experience; they also realized significant business advantages. The new foam component was more competitive in price than the previous supplier’s version and better met all quality requirements.
This approach proves that when designers consider molded foam at the onset of a project, they can simplify assembly and create a product that is both clinical-grade and home-ready.
Safety and regulatory compliance in the home
Transitioning medical devices to a residential setting introduces a new set of safety and regulatory challenges. Because these devices are working outside a supervised clinical setting, the materials used need to be inherently safe. While traditional foams provide basic padding, POLYFORM® custom molded solutions are engineered to meet the highest standards of durability and protection.
Thermal management and fire safety
During long operational cycles, internal components of home medical devices can generate significant heat. Custom POLYFORM® components can be engineered with specific thermal properties to act as a vital barrier, maintaining safe touch-temperatures on the device’s exterior to prevent accidental burns.
Fire safety is non-negotiable in a home setting. Our molded solutions are formulated to meet the UL 94 standards. This provides peace of mind without the need for harmful chemical additives that could compromise the air quality of a domestic environment.
Durability and interference
For a device to be truly clinical-grade, it must maintain its structural integrity over its entire lifespan. POLYFORM® components are specifically engineered for maximum durability, ensuring they do not flake, crumble, or shed particles that could interfere with the device’s sensitive internal mechanics.
Additionally, in our hyper-connected homes, electromagnetic interference is a constant threat. So, much like we did in our case study mentioned above, we apply a conductive fabric shield directly onto POLYFORM® molded components. This allows our products to protect medical devices’ sensitive diagnostic data from home WiFi interference, ensuring accurate readings.
Trust in our expertise
The shift toward home-based medical care presents a significant opportunity for OEMs, but only if they can master the delicate balance of power and peace. A device that meets clinical goals but fails the bedroom test due to noise, heat, or discomfort will ultimately struggle in a competitive market.
Engineers can exceed regulatory and patient expectations by using specialized materials like POLYFORM® for ergonomic support, protective housing, and flame/thermal resistance. Polymer Technologies provides integrated engineering solutions, not just foam, ensuring life-sustaining technology is durable, comfortable, and safe in the home.
Contact one of our engineers today to discuss a custom-engineered solution for your next medical device.